Biofilms that form in the human body are up to ten thousand times more resistant to antibiotics than free-floating bacteria, making them very difficult to treat medically. These biofilms are responsible for the extreme persistence of many difficult to treat illnesses like Legionnaire’s disease, Staphylococcus aureus (“Staph”), and infectious bronchitis, that can trouble patients with frustrating symptoms for years.
Some years ago researchers showed that biofilms might also be helping the Lyme-causing bacteria evade treatment.(1) These findings have excited Lyme researchers who have since been exploring various treatment strategies designed to target the entire bacterial colony. If successful, these treatments might bring long-needed relief to patients with late-stage or persistent Lyme disease where antibiotics have previously failed.
At Bay Area Lyme Foundation, we are also inspired by these discoveries and hopeful about the treatment options they may bring. Recently we invited Daina Zeng, a Senior Scientist at Agile Sciences, to talk about the work her team is doing adapting Agile’s proprietary non-toxic organic compounds to disperse these bacterial colonies (technology they have leveraged for medical, agricultural, and industrial uses). Her post follows.
What is a biofilm?
A biofilm is something that we come into contact with every day. The plaque that forms on your teeth and causes tooth decay is a type of biofilm. Clogged drains are also caused by biofilm, and you may have encountered biofilm-coated rocks when walking into a river or stream.
Biofilms form when bacteria adhere to surfaces in some form of watery environment and begin to excrete a slimy, glue-like substance that can stick to all kinds of materials including metals, plastics, medical implant materials, and biological tissues. Biofilms can be formed by a single bacterial species or many species of bacteria, as well as fungi, algae, protozoa, debris, and corrosion products. Essentially, a biofilm may form on any surface exposed to bacteria and some amount of water.
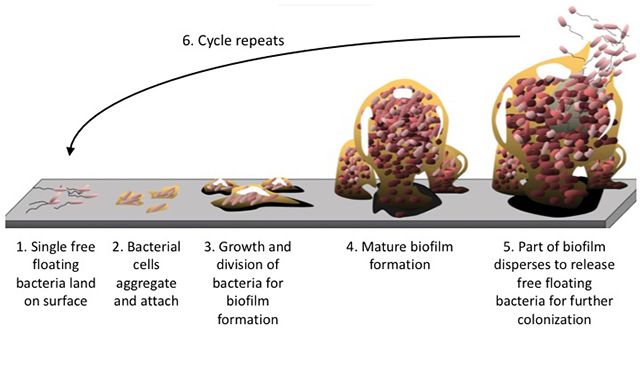
Biofilms are thought to be responsible for more than 80% of microbial diseases, such as:
- Otitis media, the most common acute ear infection in US children
- Bacterial endocarditis, infection of the inner surface of the heart and its valves
- Cystic fibrosis, a chronic disorder resulting in increased susceptibility to serious lung infections
- Legionnaire’s disease, an acute respiratory infection resulting from the aspiration of clumps of Legionnella biofilms detached from air and water heating/cooling and distribution systems
- Chronic wounds (wounds that do not heal within three months)
- Hospital-acquired infections, such as infections acquired from the surfaces of catheters, medical implants, wound dressing, or other medical devices
How might a biofilm explain the persistence of Lyme?
Forming a biofilm is a very effective protective mechanism and bacteria encased in a biofilm are highly resistant to antibiotics (up to 10,000-fold). So antibiotic treatment is often times not effective against cells protected in a biofilm. Unfortunately, cells within a biofilm can return to their freeliving form and escape to form new biofilms and/or colonize new tissues. Since antibiotics can’t be continuously given to a patient, there really isn’t anything to stop these bacteria cells from spreading the disease. It’s really a vicious cycle.
What has the research actually shown regarding Lyme disease and biofilms? What is the importance of some of the more recent studies?
Even though biofilms have been attributed as one of the underlying causes that make other chronic bacterial infections so difficult to treat, (such as cystic fibrosis, chronic wounds, endocarditis, etc.), biofilms have not been well studied in Lyme disease. There are papers showing that Borrelia burgdorferi, the causative agent of Lyme disease, forms biofilms outside of a host (in vitro).
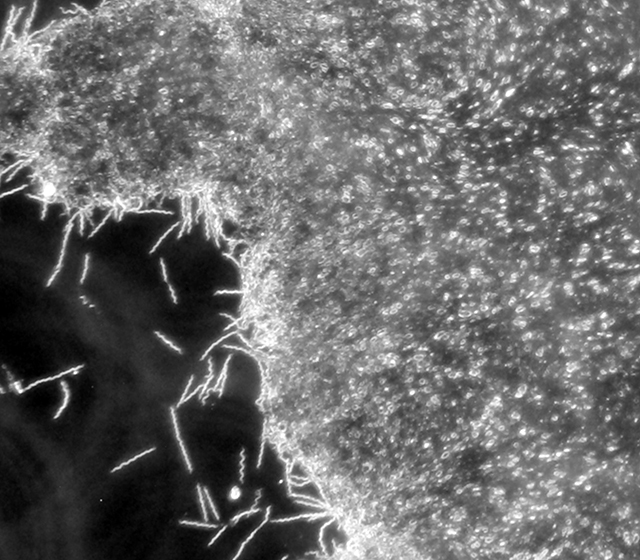
Current studies are being done to probe the presence of biofilms in animal models and even human patient biopsy samples. We look forward to the results of those studies that should be coming out in the next year or so.
These studies are very important since they increase our understanding of Borrelia burgdorferi; and given that biofilms are so prevalent and relevant in other pathogens, it’s worthwhile to study whether biofilms play a role in Lyme disease as well.
Is it possible that the antibiotics might actually trigger the biofilms to create a stronger barrier?
Absolutely! Biofilm is a protective mechanism that cells turn on when they sense they’re under stress, so there’s significantly more biofilm formation when cells are treated with antimicrobial agents including antibiotics. Numerous studies have shown this for nearly all antibiotics.
To what extent does it mask the Lyme bacteria from detection?
Since bacteria are encased in a biofilm and are not free-swimming in solution, they can be hard to detect using diagnostic tests that rely on solution samples (i.e. blood, etc).
What can be done to combat the biofilms?
The only way to effectively combat a biofilm is to “pop the bubble” and disperse it. There are two major ways to do that: (1) using mechanical force to scrape it off (similar to a teeth cleaning), or (2) tricking the cells into dispersing themselves. Since Lyme disease is within the body, it’s not possible to use mechanical force. So the only available tool left is to use chemical compounds that inhibit the ability of cells to form and maintain biofilms.
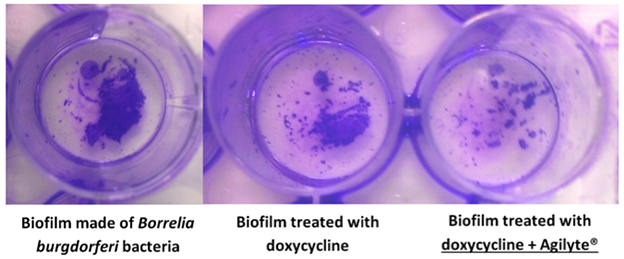
At Agile Sciences, we have developed compounds that inhibit bacteria’s ability to turn on the pathways required for biofilm formation. So when we treat bacteria with our compounds, we see significantly less biofilm formation, and we’re also able to disperse existing biofilms. We think the best use of these compounds is in combination with antibiotics. The idea would be for our compound to “pop” the biofilm so the antibiotic could now kill the pathogen.
Support from Bay Area Lyme Foundation has allowed us to identify compounds (known as Agilyte®) that have efficacy against Borrelia burgdorferi biofilms and show synergy with commonly used antibiotics in Lyme disease, including doxycycline (see Figure below), ceftriaxone, and amoxicillin. We’re currently applying for further funding from NIH to develop these compounds as a novel therapeutic to enhance antibiotic treatment of Lyme disease. See here for more information about the Agile project.
What new research might help advance this line of treatment?
At the end of the day, I really do believe we need to have a further understanding of how Borrelia acts in the human body and whether they form biofilms. Since biofilms are so prevalent and increasingly implicated in more and more diseases, I think it’s important to study if it plays a role in Lyme disease.
On our end, we’re working on getting funding that will allow us to take our compounds into animals to see if using our compound can enhance doxycycline treatment in mice.
*****
(1) Sapi et al. “Characterization of biofilm formation by Borrelia burgdorferi in vitro.” PLOS One, 2012: 7 (10).
(2) Monroe, D., “Looking for Chinks in the Armor of Bacterial Biofilms,” PLoS Biology, Vol. 5, No. 11, e307 doi:10.1371/journal.pbio.0050307
Images courtesy of Monroe (Stages of Biofilm Development), Sapi et al. (Borrelia burgdorferi biofilm), and Zeng (Agilyte® Technology).

I got bit two years ago by bugs over my entire body. I have sores that will not heal & nothing affects them. I suffer with daily dizziness, headaches, absence seizures & fatigue. I’ve tried everything to kill these parasites on my skin. Afraid I will die from this
Hey Donna,
I used Ozone, Hyperbaric Oxygen Therapy and herbs to put my Lyme into remission. Have you tried these?
So sorry for your circumstances
. I have had a to similar condition for several years now. So many doctors have me I pop
Lady the was having d5 delusions of parasites. How about you?
I read that Wild Oregano oil dissolves Lymes biofilm. I have been taking 1 to 2 drops under my tongue on and off for a year and have less symptom, of feeling like my head is in a vice. Hope this helps someone.
Thank you, it does help.
Cistus Incanus tea is completely natural andvery effective in destroying Bartonella’s biofilm. Not sure if it works with Borrellia but there are good chances. Not all brands work though. The one that works for sure is Biopure. I am not affiliated with any company or have any commercial interests. I am just a patient healing from a very severe and long lasting Bartonella infection using several potent herbal tinctures and Cistus tea.
How long have you been taking the tea and when did you start seeing results?
Hi
How do you know it works for bartonella? Have there been studies done? I’m interested in trying if there is science behind it. Biaj53@yahoo.com
Thanks
Hello
I read your comment what are the potent herbal tinctures you are using. And have you taken antibiotics?
Hi Sara
i have severe abdominal pain from Bartonella its hell in the tissue and cant get it out what was your protocol and what worked for you i hope you are better. What were the timescales and how long are you ill
Neil suffering like hell!!!!
Dear Friends ~
I just came across this Biofilm article:
“Peppermint-Bombs-Blast-Bacterial-Biofilms”:
https://cen.acs.org/articles/93/web/2015/07/Peppermint-Bombs-Blast-Bacterial-Biofilms.html
Good Luck !!
I read the article and the clinical study involving cinnamaldehyde from cinnamon and peppermint oil within colloidal buffer to destroy the biofilm…it is still under research as the research states when the bacteria are released from the biofilm antibiotics are then used to kill them. It was in 2015. I can’t find any further study information from professor rotello at Amherst university.
Hi. There is a study about biofilm being removed from Lyme Spirochetes with Cassia and Bay Leaves. They tried other stuff but these 2 got rid of 20 to 30 percent of biofilm.
The Bay Leaf (not eaten) is something to add daily to Lyme treatment. Its high content of Eugenol is the cause. There are also other research papers showing Eugenol to have an effect on other bacterium.
Cassia is kitchen cinnamon. Not great for your liver in large quantities. Ceylon Cinnamon or Cinnamon Bark Oil are easier to use.
Also for fatigue, ionic copper applied to wrist or under feet drops the spirochete out of red blood cells. It has to be ionic. The immune system then just eats it.
Stevia is also used. It unplugs the defensive mechanism Lyme uses to close the tubules above cysts. Stevia reverses this.
Also Kudzu in tablet form greatly changes the illness. It works for 4 to 6 mos, de-inflammes the CNS.
Hi Ro, thanks for the great information! Could you please provide brand of ionic copper if you don’t mind? What form of stevia are you referring to (ie tinctures,essential oil, capsules)? I had never heard of kudzu. Also do you know what is good to take for Bartonella? Thank you kindly
A cautionary note about copper. My son has epilepsy that was caused by chronic lyme disease. He has a copper overload that increases the number of seizures. He has to take a high dose of zinc to counteract this. Others with epilepsy may want avoid adding copper to their diets.
Check out FloridaSharkman.org Lyme protocol
Meds available from https://www.virex.health/index.php?route=product/product&product_id=53
Based upon peer-reviewed medical journals, Ceylon Cinnamon, Oregano Oil, Allspice Extract, and Garlic extract used in conjunction with several courses of Doxycycline is what finally provided the breakthrough cure for me after battling Lyme for nearly a decade. I truly believe it was the combination of antibiotics and the extract supplementation that finally worked for me.
https://publichealth.jhu.edu/2018/essential-oils-from-garlic-and-other-herbs-and-spices-kill-persister-lyme-disease-bacteria
https://pmc.ncbi.nlm.nih.gov/articles/PMC5641543/
In my 30+ years of using the internet daily I have written less than 10 reviews/comments, but I feel it is important to spread the awareness about what finally worked for me in case it could be helpful to someone else.
I AM NOT A DOCTOR, I CANNOT STRESS ENOUGH, DO NOT start a regimen without discussing with your DR. who will likely want to monitor your liver enzymes while taking any supplements…. even food grade ones.
My only other advice is do not give up. Find a doctor that truly cares and believes you. It can get better!
***********************************************************************************
*My review for my DR. after ~1 year of treatment:
Dr. Soni is the only doctor I trust. I suffered from malaise and fatigue daily for just shy of a decade; doctors across 2 states telling me I’m healthy, and my issues were likely psychological, and induced by stress. For my first apt. with Dr. Soni, she truly listened and discussed my entire medical history for more than an hour— for the first time in my dealings with the medical system I wasn’t just patient 2222. Over the course of a month, she test after test, until I had a diagnosis and treatment.
Also, while she was not supportive of any untested treatments, Dr. Soni did listen to my own research of John Hopkin’s successful in-vitro studies using certain food extracts to kill the bacteria causing my disease. And when I told her that I would like to begin incorporating those extracts into my diet (alongside her prescribed treatment), she discouraged me, but instead of just saying no, she was pragmatic and insisted that I have regular blood/liver levels checked if I insisted and did not listen.
After approximately 6 months of treatment, I was 95% better. Today, I am more than 1 year symptom free. And while during these pandemic times I’m more stressed than ever, I FEEL AMAZING!
Saying Dr. Soni saved my life is not a hyperbole. For almost 10 years I was a husk of my healthy self, and slowly getting worse with more symptoms. Unlike every other doctor who saw a fit and healthy patient, she listened and saw through the façade, to see a human being suffering and wanted to help.
Words cannot express the gratitude I have for Dr. Soni.
This is for Cindy. My heart goes out to you because you sound just like me. Mine started around spring of 2019. I had a tick bite and another insect bite that was infected, but I didn’t get it checked because there was no bullseye. Then the problems started. I first noticed the black dots on my back that looked like beauty marks or black heads that were never there before. I’d wake up with what looked like little bug bites. Went to drs, they said I had bed bugs. Had exterminator come and we did not have bed bugs. Kept getting worse — the intense itching, crawling under my skin, and biting started. Went to dermatologist, said I was being bitten in the garden. Went to ER, they said they couldn’t help me and I insisted on a Lyme test. They only did the basic test, it came back negative. Went to my primary dr. She did the western blot which came back positive. Was on antibiotics for over a month, they did nothing. I started losing my hair, it became dry like straw; lost tons of weight; had debris in my bed and filaments coming from my skin. Some are painful because it’s keratin and collagen breaking through, but also there could be parasites which I saw after soaking in epson salt and other things & essential oils. Since Lyme leads to co-infections, I believe I have a fungal and bacterial skin infection. I’ve researched and found that I may have something called Morgellons which some research has found is connected to Lyme disease. The filaments have the spirochetes in them. I don’t sleep until 4 and 5 am. I’m exhausted and in excruciating pain all the time. I do lots of research and am seeing a homeopathic dr that is helping me, but I also find what works for me. There is a skin debriefing soap that I buy from Richard Kuhns. Look him up. He has a call every Sunday at 4pm where people that are suffering can call in and talk to others and to him because this is a very isolating disease and can make you think you’re crazy, but you’re not. It is real. I was getting better using lugol’s 5% iodine and colloidal silver. Now I’m taking the lumbrokinase and Serrapeptase and so many other things. I try to eat raw garlic everyday and take glutathione. I’m still not 100% well, but I’m at least 90% better. At least the crawling has stopped. I’m sorry you are going through this and really hope you get this message. Research your symptoms and do what’s best for you. Like you, I have a husband that is very supportive and has been a blessing. Take care of yourself and don’t give up.
Thank you for your words! This has been so hard. I havent found a Dr. Yet, but I started taking monolaurin and garlic and silver. Im doing better. Not 100% but better. I don’t understand why these drs react and treat patients this way? They took an oath. It almost feels like there is an agenda behind it. Thanks again! 🙂 Cindy
serrapeptase to dissolve the biofilm followed up with hyperbaric oxygen to blast the spyrochetes……hyperbaric chicagoland.com
You describe classic symptomatology for Morgellons disease. I have been researching this complex disorder for 3 years and may have seen some evidence for it in a few dogs and cats. Unfortunately, most physicians including dermatology specialists dismiss everyone with your symptomatology as suffering from a psychological disorder called “delusions of parasitosis” and refer these poor suffering humans to a psychiatrist. No firm answers yet!
T J Dunn, DVM
Wow you just described what I am a month into. Im probably gonna have to shave my head and I love my hair. I need to clean but hurts when they attack me … 🙁 if you could send list of what works I would appreciate it
Have you ever tried MMS/Jim Humble? Many people with Lymes, Morgellons, etc. have amazing testimonies.
I am a long line of Mètis foragers and voyageurs; we rub our skin with ash and lemon balm/mint before entering bush to hunt. I made a TINCTURE out’ve the plant root of the tall teasel. Used daily will bust up cysts, reset your nervouse system and defend agaist future attacks
Bon chance mesamis!
I had Lyme in 2011. Went on doxy for 4 wks and was no better after that. My chiropractor suggested using red infrared laser therapy. The protocol was from a doctor in Connecticut who’s had a lot of experience with Lyme. The difference was night and day. It penetrates deep in the body and there is no concern about any biofilm. I had about 5 treatments and the Lyme was gone. Felt almost 100%, but I was re-tested by my doctor and chiropractor with kinesiology testing. So far it’s been good.
Greetings John M.,
I have just seen your post. I would like to know what protocol did the doctor recommend together with red infrared laser therapy.
Thank you.
Hi John,
Could you please tell you more about the doctor who uses infrared last therapy? Where he is located and his name so that I may call him or at least look into that kind of therapy. In all of my years of research to find treatment for my daughters with Lyme, I have never heard of this one.
Thank you so very much,
Kelly
Hi, can I ask you the name of your Chiropractor and location.
Thanks
Brian
I am very interested in the protocal you used if don’t mind sharing or the Doctor’s name. I have had Lyme for 3 years and not taking anything for the biofilm. I now have Lyme that is affecting my ear and nerves. Still looking for a doctor. Any help you can give me is so much appreciated. Getting very worried as it is in my joints also, but when it affects nervous system it is scary. Thanks again for any help.
Can you tell me your drs name in Connecticut?
Who was the doctor in ct???
Hi Jon,
Can you please tell me the name of the doctor in Connecticut who uses this red laser therapy for lyme patients? I have had lyme since 1996, no longer respond to antibiotics, recently moved to Connecticut and exploring other options so am very interested in this.
Could you share please the name and contact info of the doctor in Connecticut?
Thanks. Agnes
Why won’t you answer us?!
This is not in CT, but it may be the same thearpy John used for Lyme which apoarently dissolves biofilms:
http://lymelasercenterofcentralmissouri.com/science.html
HAS ANYONE HEARD OF CISTUS TEA TO BREAK BIOFILM? IF ANY BIOFILM BUSTER WORKS, MUST YOU BE TAKING ANTIBIOTICS TOO SO THAT WHEN THE BIO FILM EXPOSES THE BACTERIA CAN BE KILLED?
Yes, I ordered the very rare tea. It tastes awful. Worse, one has to drink great quantities of it for an extended period for it to “work.”
Try researching GARLIC benefits, how it may help. Many years ago, I watched a documentary about Lyme disease, and at the end they mention garlic. FRESH GARLIC.
SAVE THIS… Use whatever herbs you like (SPK, silver, lumbrokinase, Young Living thieves brand only, cats claw, astragalus, raw diet helps immensely no sweet food) but do frequent garlic enemas until symptoms are gone. Then do them once a week. Work with a naturopath while you do. Grate garlic in enough water for enema. Heat water until warm not hot. Cover like a tea. Steep up to 8 hrs. After declared cured by naturopath, take any conbo. Of lyme herbs as a preventive 3x day and garlic enema once a week. Lyme can hide in joints etc. And show up again for awhile. So it’s a good protocol for life. Vicks vapo rub to deter ticks. Prayer too. Help others with this method. God bless. You’re welcome 🙂
You are right. I had twenty biofilm bumps last summer so I know biofilm does not like garlic so I minced garlic and placed on big bandage and put over bump for five hours removing it and bump and skin is gone. You need to remove the skin as the biofilm knits into it. And the bandage when taken off was green. as in the war tick there is plankton and algae that hide the biofilm from your body. It worked on a biofilm rash. And if you have biofilm on your teeth, which I did on all of mine, the dentist was shocked, but that is easy, use Bragg’s Natural Vinegar, brush your teeth with it and swish it around in your mouth, it takes care of it.
Please send any new info on getting rid of biofilm on the body and add me to your list. Thank You!!
Have you heard back from anyone about any info on getting rid of biofilm?
Please send me any information you have about biofilm. I’ve been in chronic pain for over 2 1/2 years now with no relief and nothing shows up on scans and no drugs have worked either. I am working now with the distance healer that says this is probably what I’m dealing with. My chronic pain is in my lower abdomen into the pelvic floor is absolutely unbearable at times. I’ve lost all quality of life.
About 8 years ago I got the movie: Under Our Skin from the public library for free to watch and learned more from that movie than this site here. Near the end of the movie, you will see the doctor and his microscope pictures proving there was the biofilm colonies of borrelia burgdorferi that was the cause of Chronic Lyme. High strength Bountico silver ten thousand parts per million blows a hole in the biofilm and goes inside and kills the inhabitants. Do you research: Lymes ms biofilm silver
Thank you for the interest. Unfortunately the Agilyte compound, while effective against the bacteria’s biofilm, proved toxic for human consumption and is not a suitable candidate for therapeutic use. Agile and Bay Area Lyme both remain committed to exploring other promising avenues in therapeutic research. While we will inevitably encounter disappointments in the pursuit of innovative solutions, it is important that we support novel research approaches for fighting Lyme disease.
Bio-fibrin and high dose of serrapeptase.
Me too!
Add my name as well please, been dealing with Lyme for some time. But never been treated for biofilm. Thank you so much. Every nugget is so helpful when you are fighting to stay alive.
I’m presently being treated aggressively for Lyme with 2 antibiotics, malarial drug and fungal drug. I’ve taken this a month and have become very sick, nausea, headache and inability to eat. I’ve last 10 pounds in couple weeks. Is this a normal reaction? I’m also taking glutathione and now gag every time I take it plus get indigestion from it.
What about Lugol’s Potassium Iodine 2%, has that been studied? Has Lugol’s Iodine been used in the treatment of malaria? I think I had read it was more effective than the drugs. Does Lugol’s Potassium Iodine kill gram negative- and gram-positive bacteria? Is castor oil used as a carrier for some type of drug in cancer patients??? Is castor oil good on its own? Is Lugol’s Iodine effective with a combination of castor oil and Lugol’s Potassium Iodine on the skin? Believe I read that cinnamon essential oil was effective in dissolving biofilm… wonder if it is? I think it was in an iodine solution. Maybe google to investigate.
Bobby yes. Please keep going. It’s exactly how I was. Keep treating. You will get it into remission
Go to Wikipedia and read the medical term: herxheimer. Probably the most important word in the medical dictionary with only a small percentage should knows knowing its meaning.
Go to Wikipedia and type in the word: herxheimer. It is the most important word I know of in the medical industry and it and its meaning are unknown to a huge percentage of those in the industry. Just try it — ask your doctors and nurses. Do go read it for yourself. Without the understanding of this word, many people stop taking things that will help them and blame the thing they are taking for making them more ill.
HAS ANY FORM OF RADIATION ERADICATION EVER BEEN TRIED ON A ISOLATED BIOFILM?
I am working with Stevia, grown in hydroponics with silver as one of the nutrients. I would like to see if we can test this in dogs we think may have Lyme. Not yet ready to try with humans due to possible side effects of this Stevia.
Did that work?
Hello I’m a Lyme sufferer 24 years,
I suffered from Lyme disease since my childhood 24 years, and I found no solution and help in my country.
I am desperately looking for any kind of help or advice, how to fight or how to recover from this disease.
If there is any chance or solution for a treatment, or if you know a Lyme disease specialist MD in Europe, please write to me ! Please Help me!
Hi Mihaly, Unfortunately awareness and understanding about Lyme disease is only slowly growing, but it is growing and there are ways to get help. We would strongly encourage you to consult the International Lyme and Associated Diseases Society (ILADS) physician referral tool, it is an international database and may help you find a specialist in your area. You may also want to look for local Lyme disease support groups as those communities may also offer suggestions about local experts and resources. We wish you the best of health and hope you find the care you need.
There are many groups on Facebook with people who can guide you in finding help.
I understand how you feel I’ve had chronic late stage Lyme disease for over 20 years! I would recommend Dr Klinghardt. He has 3 offices in Europe-he is a Lyme Literate MD originally from Germany. Google: Sophia Health Institute that’s his website. Good Luck to you!
I am not a medical professional nor have Lyme. Also not a seller but have pylori [bacterial infection in digestive tract] ongoing. But I see some useful supplements that you can try.
1. Interfase Plus (this breaks the biofilm)
2. Monolaurin (coconut oil derived) or you can use your regular antibiotic
Caution: Always start with a very small dose and gradually increase it. Some microbes exhibit very unpleasant die off effects. I wish you good health.
Google the salt protocol. Do that for two weeks. Then do replenishing with probiotics for two weeks. Eat whole foods. Take an adaptogen like holy basil or red korean ginseng. The idea is to activate the immune system. The adaptogen will kick ur immune response up or down as needed. Rest. Take cbd oil and.or black seed oil also. This is what I did to save my little girl from lyme and it worked.
I live in the Netherlands and had Lyme for 2 years. I used Monolaurin 600 m. capsules 3 x a day for 6 months, then gradually cut down. I’m symptom-free 3 years later with only one maintenance capsule of 300 mg. per day. I did some other things as well, but this is what helped me the most.
Biofilm Defense by Kirkman.
Hi Mihaly,
I may have someone i can recommended.
Im going through Lyme myself now. Contact me back when you see this
Hello I’m a Lyme sufferer interested in your help or providing any info to assist in any way. We are desperate for relief. Thanks for your work.
Try and find an ondamed practitioner. Its a device that kills the bug in spite of biofilms. It will tell you what level your infection is and even what infections you have.
All my family is suffering of this disease, including a kid of 11 years than 10 years. I am very interested in this, please add me to your list. Thank you for your research.
Best wishes, Irina, to you and your family. We will definitely pass on your contact information to the research team.
Hi, I have been reading a lot about Lyme treatments. Have you tried Buhner’s protocol or Rawl’s protocol?
I do not live in the Bay Area but on the East coast. I’m sorry for the delay of your funding but if it gets going again, I am interested in your study if possible. If not, do you know of any similar studies out this way? Been suffering for 10 years + with little luck in busting the biofilm. Can you tell me where to find the ionic silver you speak of? Thank you!
Hi Patti, We will certainly pass on your contact information to the researchers for when the study resumes. In the interim, you may find some suggestions about how to find other studies and clinical trials from this post earlier this spring. (The last section talks about various databases that track active studies. Best wishes.
I would like to have my contact added to any future study. I am suffering greatly with most likely Babesia which is slowly draining the life and the will from me.
We will absolutely pass on your interest and contact information to the researchers and wish you health and recovery.
Silver Ions Bust Through Bacterial Biofilms. A number of new studies demonstrate conclusively that antimicrobial silver busts through bacterial biofilms, and can even prevent microbes from forming biofilms. This is important news because biofilms are the protective “fort”-like structures erected by colonies of pathogens in order to evade the effects of antibiotic drugs.
Here’s what current clinical research is demonstrating about this most remarkable quality of antimicrobial silver…
Another study published in the journal Antimicrobial Agents and Chemotherapy (Dec. 2005, American Society for Microbiology), revealed that the extracellular matrix around Staphylococci epidermis biofilms have been found to be “markedly impaired” by very low levels of silver ions. According to the study authors, silver ions destabilize the extracellular polymeric substances (i.e., EPS) of which the biofilm matrix is constructed and held together. The study authors state:
Now that’s quite a mouthful, isn’t it? But in plain English the study authors are saying, essentially, that the silver ions destabilize the biofilms through an electro-chemical process that ultimately robs the biofilms of their structural integrity. The extracellular matrix, or “fort” that’s been constructed around the bacterial biofilm colonies begin to fall apart, thereby exposing the pathogens. Importantly, the silver ions can accomplish this within 60 minutes of exposure to the biofilms, and at very low levels.
The study authors focused on two different strains of Staphylococci epidermis — a Staph bacteria of the skin that’s very similar to Staphylococcus aureus of MRSA fame. Other pathogens such as Psuedomonas aureginosa are also known to protect their colonies from the effects of antibiotic drugs with biofilms.
In another study, published in the journal Colloids and Surfaces B: Biointerfaces, (Vol. 79, Issue 2, Sept. 2010), titled “Silver Nanoparticles Impede Biofilm Formation by Pseudomonas aeruginosa and Staphylococcus epidermidis,” it was demonstrated that a staggering 95% inhibition of biofilm formation could be achieved within 24 hours simply by treating the Pseudomonas microorganism with silver nanoparticles. This may be one reason why colloidal silver is so effective against Pseudomonas infections, as well.
Plaque on teeth is also a protective biofilm produced by dental pathogens. Interestingly enough, in the past, silver has also been shown — both in animal studies and in research for a dental patent — to help rid teeth of plaque.
Even E. coli bacteria have been known to produce biofilms. And of course, E. coli is another pathogen known to be susceptible to the infection-fighting qualities of antimicrobial silver.
And in another study, published in the journal Biofouling, titled “Silver Colloidal Nanoparticles: Antifungal Effect Against Adhered Cells and Biofilms of Candida albicans and Candida glabrata,” it was found that silver nanoparticles exhibited fungicidal activity against two species of fungi “at very low concentrations.”
More importantly, it was discovered that the silver nanoparticles were effective in inhibiting biofilm formation by these fungi – in other words, the silver prevented the fungi from building biofilms. However, the researchers found that in the case of these two fungi, the silver nanoparticles were not quite as effective in breaking down existing fungal biofilm formations.
In a more recent study titled “Silver Enhances Antibiotic Activity Against Gram-Negative Bacteria,” (Science Translational Medicine, June 2013), it was revealed by the study authors that silver-treated bacteria weren’t even able to produce a biofilm at all, and that when drug-resistant biofilms were treated with silver, the bacterial resistance to antibiotic drugs was completely negated.
“Overall, what we show is that small amounts of silver, non-toxic levels, can be used in conjunction with commonly used antibiotics to treat persistent infection and to treat bio-film based infections, which are problematic for medical implants,” study author Jim Collins said.
Also quite interesting is a 2001 press release from a company called NVID International, Inc., developers of a disinfectant product called Axenohl, which is “an ionic silver-based antimicrobial technology.”
Axenohl, composed of stabilized ionic silver, was tested in the municipal drinking water system in Grecia, Costa Rica, which had become non-potable (i.e., non-drinkable) due to coliform bacteria contamination. As a result of the use of only 78 parts per billion Axenohl in the city’s water supply – a remarkably small amount of silver — the bacterial count decreased from over 200 CFU (i.e., coliform forming units) per 100 ml water to zero CFU per 100 ml water, in only 10 days. Moreover, “Axenohl proved to control and reduce the presence and growth of biofilm in the piping distribution network.”
So thanks in large part to recent research, it’s becoming abundantly clear that in many cases, silver is able to either inhibit the formation of bacterial biofilms, or even bust through and destroy existing biofilms, depending upon the type of pathogen involved. This is great news, because it demonstrates another important quality possessed by antimicrobial silver which is not possessed by Big Pharma’s failing prescription antibiotic drugs. And it offers new hope that antimicrobial silver will ultimately be recognized as the solution to the growing problem of drug-resistant pathogens.
Can I be added to any list agile is compiling ?
Yes, of course. We will pass on your contact information. Thank you for your interest.
Lyme literate MD in mexico had me do a PCR blood test. Confirmed Borrelia B + Babesia micoti co-infection. 20 yrs worth of misery. + mother died from similar “symptoms.”
Was that in Puerta Vallarta? I went to a Lyme clinic there and saw improvement.
My son has chronic neurological Lyme. He has no use of his right hand and drags his right foot. His thinking is unclear and he has to ask us to repeat information over and over. He is 26 and has regressed to a preteen in behavior. We are so saddened and frustrated with how to help him. We would be amenable to any studies with which he could participate.
Colonial Silver destroys biofilms!
Try colloidal silver and look into Rife frequency therapy using electricity as a means of destroying pathogens.
Lyme is a type of spirochete. Look up what other bacterias are spirochetes. If those bacterias use penicillin to kill spirochetes, it may be a good treatment but good luck finding a doc to agree.
It sounds like your son may have marie charcot tooth disease. Have a dentist do a genetic test for tremponema denticola.
I had the same neurological symptoms from Lyme disease. Taking tinctures prepared by herbalist Dana Wood in Manitoba. She is the one who helped me. Google her and call to help.
HI Beth, My granddaughter has Lyme and Bartonella and twice for several days, she becomes like a baby and has to be picked up and taken to toilet or bathroom to wash her. Again picked up to feed her at the table.
I sent a message to Dr. Ross, who is in Seattle and does an hour and a half atfter work taking questions from people re Lyme, so I asked him about that. He had just helped a girl with same symptoms, took a while but found out in was Bartotonella that caused that. I for a year or so had my right leg and foot go completely numb. I keep. a stiff pillow with a wide base that fits under my top leg, with high sides to support it and then pull car seat up to reach to pedals with pillow under leg. You might check and see if there is Bartonella. Hope you can remedy your son’s problems. Sharlene
Very interested in any research. Willing to be a volunteer to help diagnose Lyme. As of now I cannot receive any treatment because not enough bands show positive. I am severely ill. Kidney failure. White lesions on brain. Spine being attacked by something. Regular MDs cannot figure it out. Please help!!
You need to get to a Lyme literate MD VERY QUICKLY. My daughter only had 1-band positive and I begged for antibiotic treatment. After seeing 33 MDs we finally found someone to help. Dr. Elena Frid in NYC. I thank God for her numerous times a day. Don’t waste you time, money or health by seeing anything other than a Lyme literate MD. I am a nurse and supposedly took my 29-year-old daughter to the best doctors. Not so. Half don’t believe in Lyme disease and the labs can give 50% false negatives. God Bless you!
Sorry to hear about your doctor’s shortcomings concerning your health. As Dawn stated you need a qualified doctor that knows how to treat Lyme. The test you were given for Lyme is now obsolete and you shouldn’t rely on it. There is a large network of qualified doctors around the world that can properly treat Lyme disease and other similar ailments, along with all their co-factors and complexities. Dr. Jay Dividson is one such doctor and could be referred to as the head of that large network. Many of the doctors in the network have had Lyme themselves and in Jay’s case, his wife. Weather you use Jay or ask him for a referral, they can and will help, but don’t expect a simple magic pill. There is none!
Jenni – If you haven’t already found a Dr. to treat you – find a Lyme Specialist who will do an NK CD57 lab test – performed at LabCorp. If your numbers are lower than 60 you most definitely have Lyme disease. I tested negative for all bands on Western Blot, but my CD57 marker was 13! Normal is 360 or higher. After a few courses of antibiotics the Lyme bands started showing up. If your immune system is not producing antibodies to fight the Lyme – the bands will not show up, therefore, you cannot rely on Western Blot and ELISA alone. I’m proof of that! I’m still fighting the battle and learning more and more as I read the information posted in this type of forum/website. Stay strong and persistent and trust your instincts! I saw 6 Drs before I found one with the knowledge to test CD57 and ignore my negative Western Blot.
~Best of luck to you and BLESSINGS!
I have been fighting chronic Lyme for 7+ years. I would be interested in taking part in the study.
There are so many of us that need the right treatment for this disease. Please add my contact to the list of desperate people searching for a cure.
I have chronic Lyme. If there is a study, I am interested. Thank you.
How is this study going?
From the research team at Agile: Unfortunately the original project has been stalled due to their inability to secure additional government funding to continue the research, however, they are making headway in some related work and hope to share the results later this year. This work, while not directly in the field of Lyme disease, examines compounds with similar mechansims of action and hopefully will help with the FDA approval of the Lyme-related research and/or other funding opportunities from the government. Please stay tuned for more updates later this year. Thank you for your interest and support.
I have been in this fight since 1982. Been doing oral and IV Abx and recently looking at ways to bust biofilm. I have a PICC and am interested in ways to aggressively do this. If there is a study, I am very interested.
Thank you for your interest. We will pass on your contact info to the research team. We wish you strength in your fight and hope for a healthy future.
Biofilm is only one element of chronic Lyme disease. I regret to inform you that while Lyme can be successfully treated, it is far more complex then you appear to realize. The biofilm is not easy to correctly address, without disturbing the necessary biofilms of all the organs of the body. You must also address their present across the blood brain barrier, plus their present within the body’s cells without damaging those cells, and possible parasites that are harboring them . It is also common to have viruses, fungus, mold, multiple bacteria and parasite issues. These issues are beyond most doctors and you need a specialist. See my other comment for more info.
Sorry, but any biologically-based approach to combating biofilm is doomed to fail. The bug genome can easily develop a workaround. Try a completely new, non-biological approach: http://www.HYBENX.IT.
Simple, fast, inexpensive, safe, universal, with no absorption, no residue, no chance of resistance development.
How would Hybenx work for Lyme?
Your solution would require several facts that appear very unlikely. The plaque on teeth would need to have been the result of the same Lyme pathogens, albeit the original Lyme bacteria, not the man altered ones, and the man altered ones would have to produce the same biofilm even if harboring contributing different pathogens. Your product may be excellent for plaque, but I doubt the coincidence that it’s the proper anti-biofilm for Lyme and may even be detrimental if taken internally. Can you offer some facts to support your product’s use as an anti-biofilm for Lyme?
Does Stevia affect biofilms? Thanks.
John
Hi John, Thanks for your question. We went back to the scientists to verify but it seems that yes, Stevia has shown a surprisingly strong effect on Borrelia biofilms as is noted in this paper, “Effectiveness of Stevia Rebaudiana Whole Leaf Extract Agains the Various Morphological Forms of Borrelia Burgdorferi in Vitro“, published in the European Journal of Microbiology and Immunology last Fall. It is important to note, however, that these are in vitro results, and Stevia has not been used in animal models yet.
This is a very important distinction to make. Just because something is shown to work in a petri dish, in vitro, one does not know how it would work in the body. There is a lot of speculation about many things: oregano oil, Stevia, etc., but it’s unknown how effective it actually is, especially without reliable testing and indicators for Lyme disease and its many co-infections. My step daughter is battling chronic Lyme and we are anxious for some relief, but beware the charlatans with cures that have no basis in real science.
I would also like to added to your list… Feel free to contact and thank you for your research!
Please add my information to the list to participate in the Agilyte study. I live in the Bay area and I am very interested in this. Thank you so much.
Nicholas
We have passed on your information to the team at Agile, Nicholas. Thank you for your interest.
I am very interested in participating in a Agilyte study.
Please advise. Thanks.
Hi Marguerite, Thank you for your interest. We can pass along your information to the team at Agile so they can add you to their list for future studies.
My daughter may be interested too. Please send information to me as well?
I also would be interested in participating in the study. I have the time and no one any longer left in my life depending on me. I suffer alone day in and day out year after year. I have wounds that have not healed in six years. I peel, so very carefully, daily layer-upon-layer of film off my face but it would take a entire day to do this to my entire body. And that is just where I can reach. Anyway I am here if needed. Thank you.
Mimi, Very sorry to hear of your suffering. We will certainly pass on your information to the team at Agile. Roxie, we have also shared your information. Thank you and best wishes.
Has anyone had any luck ridding the biofilm on hard surfaces like wood floors, counter tops, cabinets, bathtubs, etc. My entire home is coated, trying to disolve it and remove it.
Cindy
Dolores, would that maple syrup could cure Lyme! Ha! Sounds like we’re still years away from tackling this disease I’ve had for more than a decade.
And, Author: “synergy” is a non-word foisted upon us by Madison Ave. The real word is “synergism” (you know, the one whose adjectival form is “synergistic” [not “synergetic”].
I got diagnosed with Lyme in June 2016. I am sorry to hear about your ten years of fighting Lyme. What is the method of treating you are doing?
HELLO, my name is Cindy, I live in Marysville Wa. Its about 4:30am, another night of not being able to sleep. I am somewhat new to all of this. I am hoping someone here can offer me some advice or maybe a good resource.
I am 52 years old and in the 1st week of June 2019, I developed a super red rash on my face. Dr said it was staph infection. A few weeks later my staph never healed completely. It changed to rash with sores that looked like bad acne, however it was NOT acne, plus I had some sort of parasite mainly on my face. I went back to the walk-in clinic – which was the beginning of Dr. Nightmare. Dr said I couldn’t have parasite on just my head. It would be all over my body. He then said my face issue was acne, (not) and my arms was eczema (not)… Dr sent me home to suffer the itching and biting, and instructed me to moisturize and apply acne meds and go to dermatologist who is 4 months out for scheduling appointments. One week later, I go back for 3rd and final time, begging them to just look at my skin or take a sample for lab test. They were rude, treated me like I was a crack addict, refused to look or test, and sent me home with referral (after they said “And if we test and find something, what would you have me do Cindy?”. My mouth fell to the floor. I was like really? They even tried to convince my husband to get me into rehab. I do not drink or do drugs, and rehab won’t fix my parasite issue.
So now months later, and a few more new drs who read my history and all won’t look at my skin or do anything BUT refer to derm, I made a appointment. In august. But 1st available appointment is in January 2020. I have battled mites all over my body, pin worms, and am still battling a couple mystery parasites. I can’t work, am fatigued, have brain fog, can’t sleep, am having memory issues, possibly hallucinations, weight loss, and I can see really big worms under my skin, moving in my feet. Real long. They hurt when they move. My ears ring, my body vibrates off and on, dizzy sometimes, fibers and weird stuff coming out of skin, hair loss. …. The list is long.
I feel I need help, but don’t know where to find it. I sometimes feel like whatever this is, it’s going to kill me. My husband is worried and yet he does not want me to go to drs anymore cuz they are not listening to me. My son thinks im over exaggerating and is not sold on the Lyme disease. I am pretty sure it is lyme. I have resorted to self treatment. I have been on fenben and reishei spore oil. I started fenben at 100mg per day, and have worked up to 300mg per day. I have seen pinworm and strongyloids (?) Die off, but none of the big guys yet.
This is a condensed version of my summer and fall this year…sorry for the long post. I’m thinking maybe go to hospital, but can’t deal with another dr. not doing his job, I will go mental if that happens again… lol… so I am looking for suggestions, advice, opinions…. ANYTHING would be much appreciated.
Looking forward to getting acquainted with you all. 🙂
We have a niece with cystic fibrosis, and battling Lyme since last spring I can say undoubtedly there are similarities. It’s as if common nagging allergies come close to being lethal when fighting lyme. Choking is a daily occurrence. I’m pretty sure my windpipe must resemble the bottom of a derelict boat at times. I have recently had progress with colloidal silver. Don’t know how long I can keep drinking it… But it is helping.
Thanks so much for this research. We need to tackle this world wide epidemic and Borrelia is but one of the pathogens injected from ticks. We also need to address the other pathogens as well and figure out biofilm formation in them. The Lyme community is currently using many biofilm busters like terminalia chebula, cinnamon oil, nattokinase, serrapeptase, curcumin, lumbrokinase, maple syrup, xylitol, and lactoferrin. Next generation DNA sequencing is what we need to ascertain ALL of the bacteria, protozoans, viruses and parasites injected into people. Again, thanks so much for this valuable research!