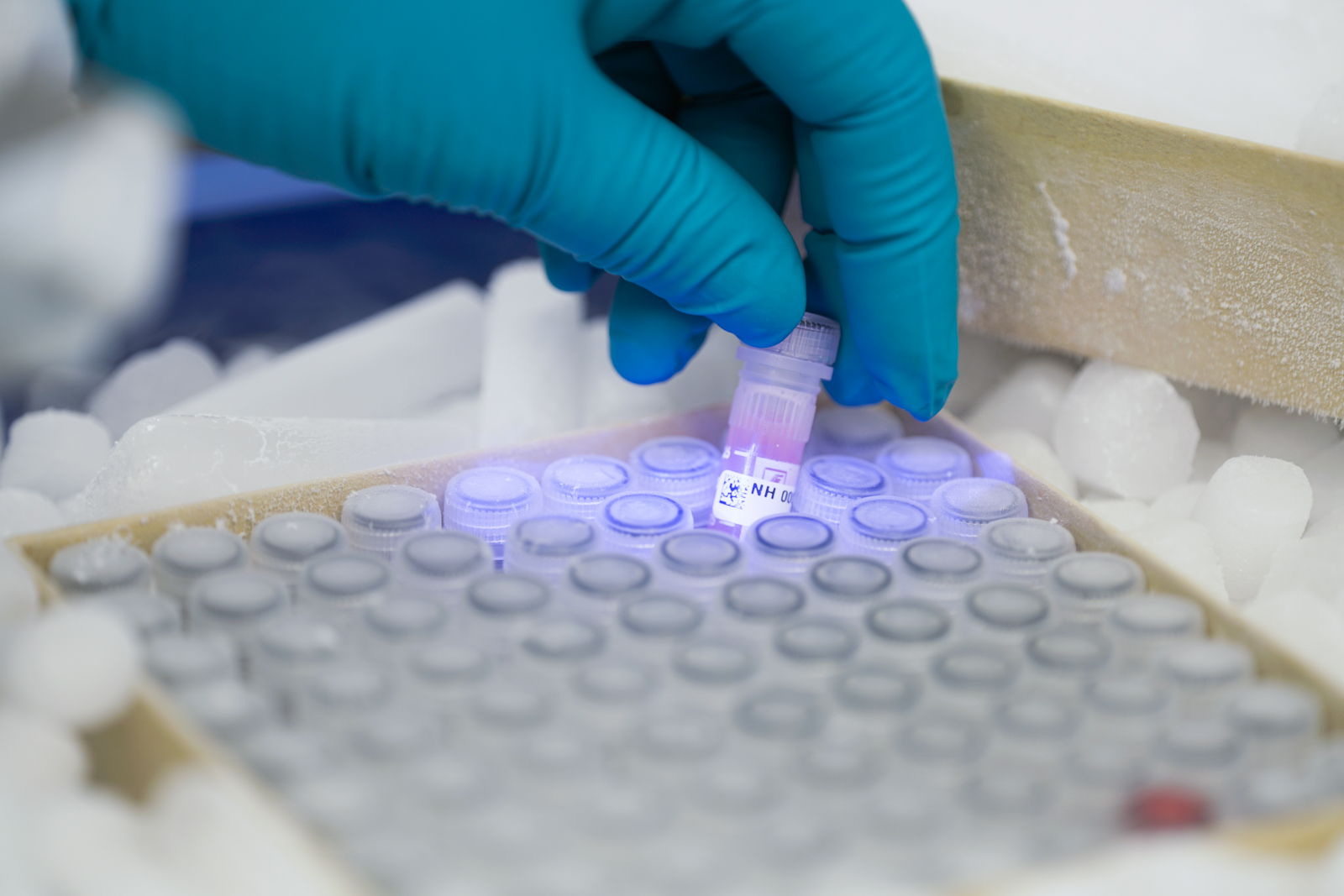
Lyme Disease Biobank® has enrolled more than 1,250 participants with early Lyme disease, later Lyme disease, persistent Lyme, or controls from endemic areas. Each participant’s donation provides samples for 125+ research projects. Our Biobank supports these projects in academia and industry with more than 25,500 aliquots distributed to date. The Biobank also collects post-mortem and surgical tissue samples.
Available Samples
Whole Blood (1 and 2 ml aliquots), Plasma (250 μl aliquots), Serum (250 μl aliquots), Urine (1 and 5 ml aliquots)
- Early Lyme disease: We have a unique cohort of samples from patients diagnosed with early Lyme disease, defined clinically, and confirmed by laboratory testing. Characterization of 550 samples collected on the East Coast and the Upper Midwest was published in 2020. Samples are typically provided blinded to investigators (not knowing which samples are cases and which are controls). An unblinded panel is available with 5 laboratory confirmed acute Lyme disease cases, 3 with convalescent draws, and 7 matched endemic controls. The unblinded panel includes all clinical data and testing data up front and is suitable for projects early in development. Previous testing in human samples is not required to access the panel.
- Later Lyme disease and other tick-borne infections (TBI): In partnership with Massachusetts General Hospital, we have collected samples from patients with Lyme carditis and neuroborreliosis with appropriate carditis or neurologic controls, as well as samples from patients with Anaplasma, Babesia, or other tick-borne infections with matched controls (with fever, high LFTs, often low platelets). We also have samples in the early Lyme cohort that tested positive for Babesia or Anaplasma by PCR.
- Persistent Lyme: We have collected ~150 samples from patients with ongoing symptoms of Lyme disease in Northern California (Santa Rosa and San Rafael) and San Diego.
Investigators interested in applying for blood, serum, or urine samples should email Liz Horn, PhD, MBI, to discuss your project and sample needs.
Tissue Samples
Please contact Liz Horn, PhD, MBI, for more information about what tissue samples are available.
Application Process
Applicants submit an application that includes a technical proposal, a lay summary of the work, justification for the number of samples being requested, timing and funding of the proposed work, the publication plan, and applicant’s CV. Applications can be reviewed without Institutional Review Board (IRB) approval, however IRB approval or exemption from the applicant’s IRB is required prior to releasing samples. Please note: There is a separate application for the Unblinded Panel that requires less detail.
Please contact Liz Horn, PhD, MBI, for the application and instructions.
Peer Review
Applications for blinded samples undergo peer review by an ad-hoc committee of at least two experts.
Final Approval
All applications are presented to Lyme Disease Biobank Board for approval. Applicants will be notified by email when approved or declined.
Signed Paperwork
De-identified samples are released upon receipt of an executed Sample and Data Use Agreement and approval or exemption from the applicant’s IRB.
Contact Us
For more information, please contact Liz Horn, PhD, MBI, Principal Investigator.
We would like to recognize and thank the Steven & Alexandra Cohen Foundation, the LaureL STEM Fund, an anonymous donor, and the 2014 & 2018 LymeAid® Fund-a-Need supporters who have contributed significantly to our Lyme Disease Biobank.
We are grateful to the participants who volunteered and donated samples.