Written by: Wendy Adams, Research Grant Director & Advisory Board Member, Bay Area Lyme Foundation
Unless you’ve been living under a rock for the last few years, you’ll have learned that tick-borne diseases are on the rise across the United States. Many theories exist as to why this is the case. However, most scientists that study ticks and their habitats agree that a combination of reasons—including climate change and human encroachment into tick habitats—are at least partially to blame.
Although Lyme disease (caused by the bacterium Borrelia burgdorferi) is the most common disease that humans acquire from tick bites, ticks can unfortunately transmit several other bacteria, viruses, and parasites to humans. Multiple infections can even be transmitted during the same bite. The confusing and overlapping disease symptoms caused by multiple infections makes it extremely difficult for doctors to recognize, diagnose and treat the different infections.
Borrelia burgdorferi and Babesia
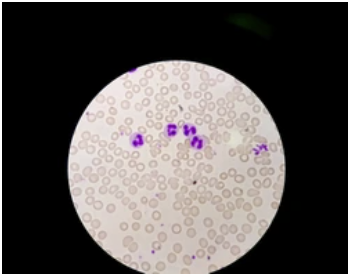
The most commonly reported tick-borne disease after Lyme disease is babesiosis, caused by infection with the parasite Babesia. This parasite, just like its cousin, Plasmodium falciparum (malaria), infects red blood cells and can cause anemia, thrombocytopenia (low platelets) and other serious, sometimes life-threatening symptoms. Babesia microti, the most common infectious parasite in the US, is typically found where Lyme disease is found—the Northeast, Mid-Atlantic, and Midwest, although most reported cases come from the Northeast. But why the overlap in this geography with Lyme disease? It turns out that the presence of B. burgdorferi in an ecosystem facilitates the establishment of Ba microti in ticks. This means that ticks in locations infected with Babesia are also more likely to be co-infected with B. burgdorferi. At least one study in mice co-infected with Babesia and Borrelia showed that Babesia hindered the acquired immune response, including B and T cell production, such that Borrelia (Lyme) symptoms were enhanced. Notably, Borrelia can affect the innate immune system—meaning that a co-infected individual might not be able to mount a sufficient immune response at either stage to fight either pathogen optimally.
Babesia is of special concern to doctors and scientists for a few reasons. Like Borrelia, the parasite can be transmitted by an infected mother across the placenta to an unborn child. The resulting infection can be dangerous in a newborn and has been documented in a case where the pregnant mother was not tested for babesiosis despite a Lyme diagnosis. Another reason Babesia is concerning is its invasion of the blood supply. Babesia can survive blood banking conditions and hundreds of cases have been documented through blood transfusions. Infected organ transplantation has also led to Babesia transmission and infection by this route is especially problematic. Transplant patients must take immunosuppressants in order not to reject the new organ, leaving them even more susceptible to opportunistic infections.
Babesia duncani—“Left Coast” Babesia?
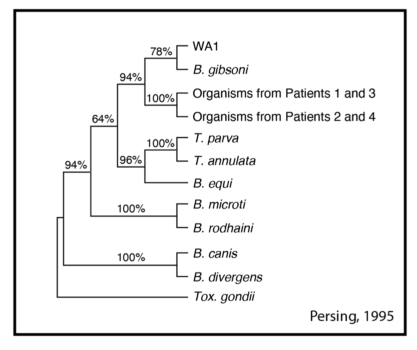
As noted earlier, Babesia is a specific family or “genus” of pathogens and there are different species within the genus in different regions of North America. The West Coast of the US has at least one species which infects humans: Babesia duncani (Babesia divergens has also been found in Washington State). B. duncani doesn’t seem to be closely related to B. microti, but instead is more closely related to a Babesia gibsoni, a species found in canines, and a parasite called Theileria. Although there are very few reported cases of B. duncani, some research suggests that it has been found in human infections in other North American locales as well and there are transfusion-associated cases in the literature. Interestingly, the strains from California—while looking the same under the microscope—differ in genetic sequence from other known Babesia strains (notably, a strain called WA1) on the West Coast. Therefore, there is significant genetic diversity even within the same B. duncani species.
Seroprevalence studies (which measure antibodies from a large population of blood serum samples) done in the 1990s showed that 3% of people in a Lyme-endemic community in Mendocino county had antibodies, and 16% of soldiers stationed at Fort Ord in Monterey county had antibodies to B. duncani. Yet, strangely, despite the overlap in regionality of B. duncani and Lyme Borrelia in California, Oregon and Washington, B. duncani has not been found in Ixodes pacificus (western blacklegged) ticks, which are the ticks that harbor and transmit B. burgdorferi. Scientists have found one tick, Dermacentor albipictus, the winter tick, that harbors B. duncani. D. albipictus is not known to bite humans often, instead spending almost their whole life on large mammals like deer or moose. These ticks also have been found to parasitize horses, cats and dogs in different parts of the country.

Clinically in humans, B. duncani can be very serious. It has been difficult to even study this species because the parasite could not be grown in a lab setting until very recently, and lab-infected mice and hamsters die within a few weeks of being infected. The first human cases included two patients who died of the infection, yet most doctors on the West Coast have never heard of it.
Clinically in humans, B. duncani can be very serious. The first human cases included two patients who died of the infection, yet most doctors on the West Coast have never heard of it.
New studies show that B. duncani has acquired resistance to regular antiparasitic drugs including atovaquone, azithromycin, clindamycin, and quinine. But recent in vitro studies sponsored by Bay Area Lyme Foundation have shown that an herbal compound studied and used in Africa against malaria (cryptolepis sanguinolenta) has shown positive treatment effects against both B. duncani and B. burgdorferi in in vitro studies. Some physicians are already using this herbal medicine in their patients in the US. (Please note: this herb has shown some toxicity and should not be taken without physician supervision)
Doctors Need to Suspect Additional Tick-Borne Infections—Many Do Not
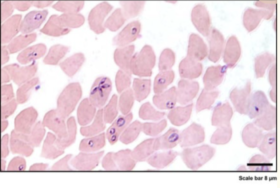
While Babesia has only been a CDC-reportable disease for a decade, we do know it is the most commonly reported tick-borne infection after Lyme disease. Despite that knowledge, babesiosis is typically not discovered until later, if at all. Obviously when a patient is infected with both a bacterium and a parasite, multiple drugs are usually required for effective treatment. Research from the MyLymeData patient registry confirms the finding that Babesia is the most common co-infection among chronically ill patients. Prompt and complete diagnosis of patients with tick-borne infections—bacteria, viruses and parasites, is paramount to giving patients the best chance at full recovery.
While Lyme disease is the most common tick-borne infection, there are others that can also cause congenital infections, severe disease and even death. Although Babesia testing is somewhat better than Lyme (a pathologist can actually see a Babesia parasite in red blood cells on a blood smear), it requires the doctor to suspect an additional tick-borne infection and order the blood smear analysis by the pathologist, which many do not. Until we have more sensitive testing, (preferably unbiased, direct pathogen testing which detects ALL tick-borne infections), and more targeted, effective therapeutics, thousands of cases of Lyme and co-infections, including Babesia, will be misdiagnosed yearly, with sometimes fatal consequences.
***
About Wendy Adams, Research Grant Director & Advisory Board Member, Bay Area Lyme Foundation

As Research Grant Director and member of the Advisory Board, she leads our research grant strategy. Wendy is also an active member of the Board of Directors for the Lyme Disease Biobank. Her unique perspective and knowledge as a former Lyme patient—as well as her background in product development and business strategy for companies like Cowen, Genentech and Aviron—make her skillset valuable to BAL. Wendy has spent over 20 years in the biotechnology field and most recently served as Chief Business Officer at Full Spectrum Genetics, Inc., an antibody engineering company pursuing programs in immuno-oncology and autoimmune disease. Previously, she ran B2DC LLC, a business development consultancy to biotech clients in infectious and autoimmune disease, therapeutic and prophylactic vaccines, oncology, drug delivery and neurology. Wendy holds an MBA from the Haas School of Business, University of California at Berkeley, where she also served on the faculty in Entrepreneurship, and an AB from Duke University. She served as a member or subcommittee member of the HHS Tick Borne Disease Working Group Federal Advisory Committee from 2017–2020 and currently serves as a Programmatic Reviewer for the Department of Defense Congressionally Directed Medical Research Program in Tick-Borne Disease. She also serves on the Lyme Disease Advisory Committee for the California Department of Public Health.
About Bay Area Lyme Foundation
Bay Area Lyme Foundation, a national organization committed to making Lyme disease easy to diagnose and simple to cure, is a leading public charity sponsor of innovative Lyme disease research in the US. A 501c3 non-profit organization based in Silicon Valley, Bay Area Lyme Foundation collaborates with world-class scientists and institutions to accelerate medical breakthroughs for Lyme disease. It is also dedicated to providing reliable, fact-based information so that prevention and the importance of early treatment are common knowledge. A pivotal donation from The LaureL STEM Fund covers overhead costs and allows for 100% of all donor contributions to Bay Area Lyme Foundation to go directly to research and prevention programs. For more information about Lyme disease or to get involved, visit www.bayarealyme.org or call us at 650-530-2439.
Citations:
Abraham, Amanah, et al. “Establishment of a Continuous in Vitro Culture of Babesia Duncani in Human Erythrocytes Reveals Unusually High Tolerance to Recommended Therapies.” Journal of Biological Chemistry, vol. 293, no. 52, 2018, pp. 19974–19981., https://doi.org/10.1074/jbc.ac118.005771.
“CDC – Babesiosis.” Centers for Disease Control and Prevention, 31 Mar. 2020, https://www.cdc.gov/parasites/babesiosis/index.html.
Djokic, Vitomir, et al. “Protozoan Parasite Babesia Microti Subverts Adaptive Immunity and Enhances Lyme Disease Severity.” Frontiers in Microbiology, vol. 10, 2019, https://doi.org/10.3389/fmicb.2019.01596.
Duncan, Kathryn T., et al. “Recent Reports of Winter Tick, Dermacentor Albipictus, from Dogs and Cats in North America.” Veterinary Parasitology: Regional Studies and Reports, vol. 22, 2020, p. 100490., https://doi.org/10.1016/j.vprsr.2020.100490.
Handel AS;Krugman J;Hymes S;Inkeles S;Beneri C; “A Case of Relapsed Vertically Transmitted Babesiosis.” Journal of the Pediatric Infectious Diseases Society, U.S. National Library of Medicine, https://pubmed.ncbi.nlm.nih.gov/32964924/.
Persing, David H., et al. “Infection with a Babesia-like Organism in Northern California.” New England Journal of Medicine, vol. 332, no. 5, 1995, pp. 298–303., https://doi.org/10.1056/nejm199502023320504.
Renard, Isaline, and Choukri Ben Mamoun. “Treatment of Human Babesiosis: Then and Now.” Pathogens, vol. 10, no. 9, 2021, p. 1120., https://doi.org/10.3390/pathogens10091120.
Research, MyLymeData Lyme Disease. “MyLymeData Chart Book Released – Information about Patients with Chronic Lyme Disease That Was Previously Unknown.” LymeDisease.org, 28 Feb. 2021, https://www.lymedisease.org/mylymedata-lyme-disease-research-report/.
Sanders, Lisa. “He Passed out Three Times in 10 Days. What Was Wrong?” The New York Times, 26 Aug. 2021, https://www.nytimes.com/2021/08/26/magazine/lyme-disease-babesia-tick.html.
Swei A; O’Connor KE; Couper LI; Thekkiniath J; Conrad PA; Padgett KA; Burns J; Yoshimizu MH; Gonzales B; Munk B; Shirkey N; Konde L; Ben Mamoun C; Lane RS; Kjemtrup A; “Evidence for Transmission of the Zoonotic Apicomplexan Parasite Babesia Duncani by the Tick Dermacentor Albipictus.” International Journal for Parasitology, U.S. National Library of Medicine, https://pubmed.ncbi.nlm.nih.gov/30367862/.
“Western-Blacklegged Tick.” TickEncounter, https://web.uri.edu/tickencounter/species/western-blacklegged-tick/.
Zhang, Yumin, et al. “Botanical Medicines Cryptolepis Sanguinolenta, Artemisia Annua, Scutellaria Baicalensis, Polygonum Cuspidatum, and Alchornea Cordifolia Demonstrate Inhibitory Activity against Babesia Duncani.” Frontiers in Cellular and Infection Microbiology, vol. 11, 2021, https://doi.org/10.3389/fcimb.2021.624745.
Harry E. Prince, et al. “Comparison of the Babesia Duncani (WA1) IGG Detection Rates among Clinical Sera Submitted to a Reference Laboratory for WA1 IGG Testing and Blood Donor Specimens from Diverse Geographic Areas of the United States.” Clinical and Vaccine Immunology, 1 Nov. 2010, https://journals.asm.org/doi/10.1128/CVI.00256-10.

Extremely well written paper devoted to different co infections and identification with Lyme disease. I have had this wicked bacterial infection including the co infections of bsbesia, bartonella, erlichia, biorrelia and others, since initial bite in 1979 in the backyard of our Scarsdale NY house. I was patient with over 20 plus doctors who hadn’t a clue what was wrong with me. This all took place mostly in Westchester county NY and I traveled to NYCity for additional commentaries. I had everything from soft inner ear bones to marital discord. FYI- still married to same gentleman for over 50 years. We are now in Florida and I have digressed back to the Dark Ages. The eminent physicians here declare that Lyme does not exist here; admit it might be something else and also have the stupidity to admit to reading a paragraph or two when in a conference and many times I am refused simple PCP treatment because ‘ I’m a complicated case.
It should be investigated to the innumerable amt of crooks in most “non Lyme” towns who are taking thousands of dollars promising a cure and prescribed meds and supplements to break the bank along with their non insured fees. It’s disgraceful and the small group of people I’ve met here have admitted they know something is wrong with them but their doctors continue referring out to specialists.
• Wendy, majority of us are neuro cognitive lyme disease / adhd patients who have lost our reading comprehension.
•
• Please have shorter paragraphs double-spaced so we are able to read and comprehend.
•
• I’ve taken 1 long, continuous paragraph of yours and broken it up for folks like me, 53 years chronic lyme/bart.
•
• I was misdiagnosed for 35 years by 40-50+ drs. losing my reading comprehension.
•
• Thank you for your consideration and even the possibility of your editing what you have done so ALL tick-borne patients are able to read your thorough articles.
•
• Betty Gordon, Ames, Iowa
• ***************************
• The most commonly reported tick-borne disease after Lyme disease is babesiosis, caused by infection with the parasite Babesia.
•
• This parasite, just like its cousin, Plasmodium falciparum (malaria), infects red blood cells and can cause:
•
• anemia,
• thrombocytopenia (low platelets)
• and other serious, sometimes life-threatening symptoms.
•
• Babesia microti, the most common infectious parasite in the US, is typically found where Lyme disease is found:
• the Northeast,
• Mid-Atlantic,
• and Midwest, although most reported cases come from the Northeast.
•
• But why the overlap in this geography with Lyme disease?
•
• It turns out that the presence of B. burgdorferi in an ecosystem facilitates the establishment of Ba microti in ticks.
•
• This means that ticks in locations infected with Babesia are also more likely to be co-infected with B. burgdorferi.
•
• At least one study in mice co-infected with Babesia and Borrelia showed that Babesia hindered the acquired immune response, including:
•
• B and T cell production, such that Borrelia (Lyme) symptoms were enhanced.
•
• Notably, Borrelia can affect the innate immune system.
•
• Meaning that a co-infected individual might not be able to mount a sufficient immune response at either stage to fight either pathogen optimally.